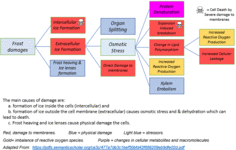
When foliage has just started growing, it's not yet producing enough sugars to resist ice crystal formation. Because the foliage is burning more than it's producing.
Add to that the fact that the cuticle is still forming so things can pass more freely through the leaf itself instead of bouncing off of a wax shield.
The more water, the harder the freeze burn.
In spring time, most plants don't produce the chemicals that reduce freezing effects, like glycerole and sugars. They use those instead to fuel growth. In general, this effect is stronger in deciduous plants.
A layer of water on the outside however, seems to have a protective effect for some reason. I don't know the exact mechanism but I think it has to do with the way water freezes and how it has to lose heat to become ice. Without solutes, it's a lengthy process to lower the temperature of water below 0°C. A thin film of ice is also a poor conductor of energy so cooling something through a sheet of ice is difficult.
You can try this effect yourself by putting a bottle of water in the fridge for a day, then boil some water. Pour the two in a glass and when you drink it right away, you can actually feel how hot and cold water don't mix themselves.